Essentially the First Law of Thermodynamics Says That
In other words it takes twice as much force to stretch or compress a spring twice as much. However not all the energy is converted into the desired form of energy such as light.

The First Law Of Thermodynamics Biology For Majors I
It was British physicist Ralph H.
. 16 Full PDFs related to this paper. Full PDF Package Download Full PDF Package. It is dangerously easy to confuse thermodynamic quantities like free energy with kinetic ones like.
The law was formulated by Josef Stefan in 1879 and later derived by Ludwig Boltzmann. The StefanBoltzmann law also says that the total radiant heat energy emitted from a surface is proportional to the fourth power of its absolute temperature. Download Full PDF Package.
The law essentially describes a linear relationship between the extension of a spring and the restoring force it gives rise to in the spring. Essentially he pointed out that the height a moving body rises is equal to the height from which it falls and used this observation to infer the idea of inertia. The law while very useful in many elastic materials called linear elastic or Hookean materials doesnt apply to every situation and is.
Borgnakke Sonntag Fundamentals Thermodynamics 7th txtbk. The second law of. The remarkable aspect of this observation is that.
Thermodynamics should as Professor Zare noted really be called thermostatics Thermodynamics is not about things moving and changing but instead about how stable they are in one state versus another while kinetics is about how quickly or slowly species react. In modern language as conservatively converting potential energy to kinetic energy and back again. A short summary of this paper.
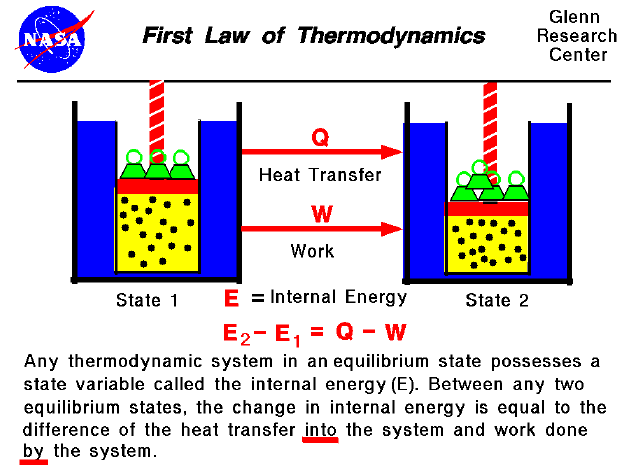
The First Law of Thermodynamics The total energy of any system and its surroundings is conserved. Having accepted the preceding axiom and associated definitions of a system and its surroundings one may state the first law of thermodynamics concisely as a second axiom. The first law of thermodynamics is simply an expression of the conservation of energy principle and it asserts that energy is a thermodynamic property.
These two axioms cannot be proven nor can they be expressed in a simpler way. The formula E σT 4 is given where E is the radiant heat emitted from a unit of area per unit time T is the absolute temperature and σ. This law first proposed and tested by Émilie du Châtelet means.
This may suggest that we could always convert energy to whatever forms we need without ever worrying about using up our energy resources. This law was understood without being explicitly stated through much of the history of thermodynamics study and it was only realized that it was a law in its own right at the beginning of the 20th century. Book - Fundamentals Of Thermodynamics 8th edition.
The First Law of Thermodynamics states that energy can neither be created nor destroyed it can only be converted from one form to another. Fowler who first coined the term zeroeth law based on a belief that it was more fundamental even than the other laws.
What Is The 1st Law Of Thermodynamics Quora

Ppt Laws Of Thermodynamics Powerpoint Presentation Free Download Id 2493855

First Law Of Thermodynamics Examples Law Of Conservation Of Energy Video Lesson Transcript Study Com
Difference Between First And Second Law Of Thermodynamics

First Law Of Thermodynamics Equations Limitations Examples

First Law Of Thermodynamics Equations Limitations Examples

The First Law Of Thermodynamics Work Heat And Energy By Matthew Gliatto Illumination Medium

First Law Of Thermodynamics In Physics Limitations Applications

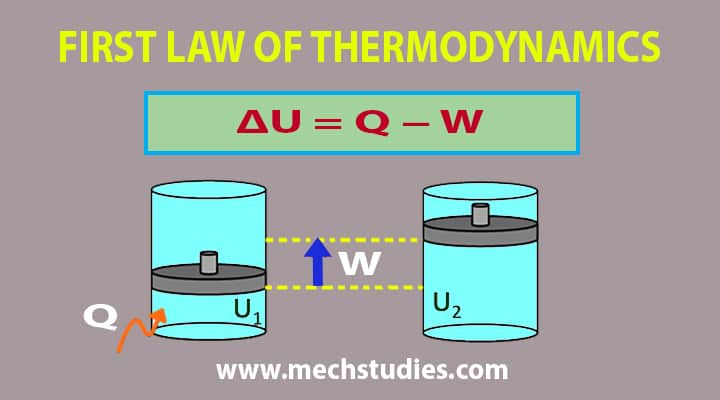
What Is The First Law Of Thermodynamics Definition Example Equation Mechstudies Com
What Is The 1st Law Of Thermodynamics Quora

Chapter 2 The First Law Of Thermodynamics For Closed Systems Thermodynamics

Chapter 12 The Laws Of Thermodynamics First Law Of Thermodynamics The First Law Of Thermodynamics Tells Us That The Internal Energy Of A System Can Be Ppt Download
What Are The First 2 Laws Of Thermodynamics How Are They Used Quora

The Laws Of Thermodynamics Article Khan Academy

First Law Of Thermodynamics Basic Introduction Internal Energy Heat And Work Chemistry Youtube

First Law Of Thermodynamics Introduction Video Khan Academy


Comments
Post a Comment